| Heading Tag | Heading Text |
| H2 | The "White Crust" Nightmare: What Just Happened? |
| H2 | The Science Behind the Spill: Why Good Batteries Go Bad |
| H3 | Reason 1: The Self-Cannibalizing Can (Carbon-Zinc Issues) |
| H3 | Reason 2: The Pressure Cooker Effect (Gas Buildup) |
| H3 | Reason 3: The "Parasite" Effect (Leaving Dead Batteries In) |
| H2 | How to Clean Battery Corrosion (Without Ruining Your Device) |
| H3 | Safety First: Gear Up |
| H3 | The Chemistry Hack: Vinegar vs. Baking Soda |
| H2 | Prevention 101: How to Stop Leaks Before They Start |
| H3 | The Golden Rule: Use the Right Battery for the Job |
| H3 | Storage Matters: Keep Them Cool and Dry |
| H2 | The Hi-Watt Difference: Engineering Against Leaks |
| H3 | Better Seals, Stronger Shells |
| H2 | Final Thoughts: Don't Let a Cheap Battery Cost You an Expensive Device |
The "White Crust" Nightmare: What Just Happened?

We have all been there. You reach for the TV remote that hasn't been used in a while, or you dig out an old flashlight for a camping trip. You flip the switch... and nothing happens. You pop open the battery compartment, and your heart sinks. The inside is covered in a flaky, white, crusty substance that looks like sophisticated snow. The battery springs are rusted, and the device looks ruined.
This is battery leakage, and it is the number one enemy of portable electronics. But here is the burning question: Why does this happen? Is it just bad luck, or is there a scientific reason? And more importantly, as a consumer or a business buyer, how can you prevent it?
At Hi-Watt, we believe that understanding your power source is the key to safety. Today, we are going to dive into the chemistry of the leak, teach you how to save your gadgets, and explain why choosing a high-quality manufacturer makes all the difference.
The Science Behind the Spill: Why Good Batteries Go Bad
Batteries look like simple metal cylinders, but inside, they are complex chemical reactors. When a battery leaks, it isn't "sweating"; it is physically failing to contain the chemical reaction inside. The causes differ slightly depending on whether you are using Carbon-Zinc or Alkaline batteries.
Reason 1: The Self-Cannibalizing Can (Carbon-Zinc Issues)
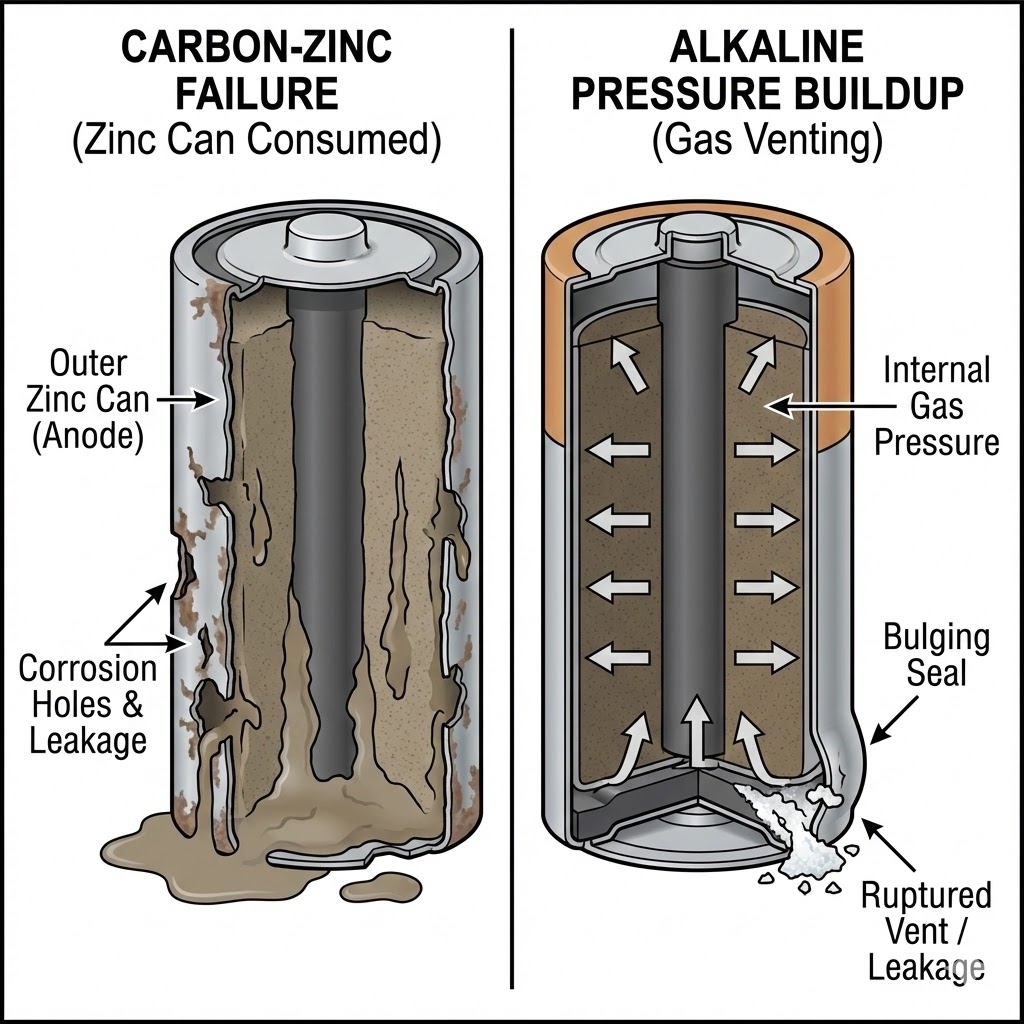
Traditional Carbon-Zinc batteries (often labeled "Heavy Duty") have a unique design flaw. The outer metal container is actually made of zinc, and it serves as the anode (negative terminal). This means the container itself is fuel for the battery.
As the battery discharges and produces electricity, the zinc casing is chemically consumed. It literally gets thinner and thinner. If the battery is used until it is completely dead, or left in a device for years, the casing can wear through completely, creating microscopic holes. Once that barrier is breached, the acidic electrolyte inside flows out, destroying whatever it touches.
Reason 2: The Pressure Cooker Effect (Gas Buildup)
Alkaline batteries are structured differently and are generally much safer, but they aren't immune to leakage. Inside an alkaline battery, the chemical reaction produces hydrogen gas.
High-quality manufacturers like Hi-Watt engineer batteries with sophisticated vents to release this gas slowly and safely. However, if a battery is exposed to extreme heat, or if you mix old and new batteries (a big no-no!), the internal pressure can build up faster than the vent can handle. Eventually, the seal ruptures to prevent an explosion, and the electrolyte (Potassium Hydroxide) leaks out.
Reason 3: The "Parasite" Effect (Leaving Dead Batteries In)
This is the most common cause of "Death by Leakage." Even when a device is turned off, it often completes a circuit that draws a tiny amount of current (parasitic drain). If you leave dead batteries in a toy or remote for months, the chemical reaction continues in an unstable state, drastically increasing the risk of a rupture.
How to Clean Battery Corrosion (Without Ruining Your Device)
So, you found a leak. Is your device trash? Not necessarily. That white crust isn't actually battery acid (in alkaline batteries)—it's potassium carbonate. Here is how to clean it safely.
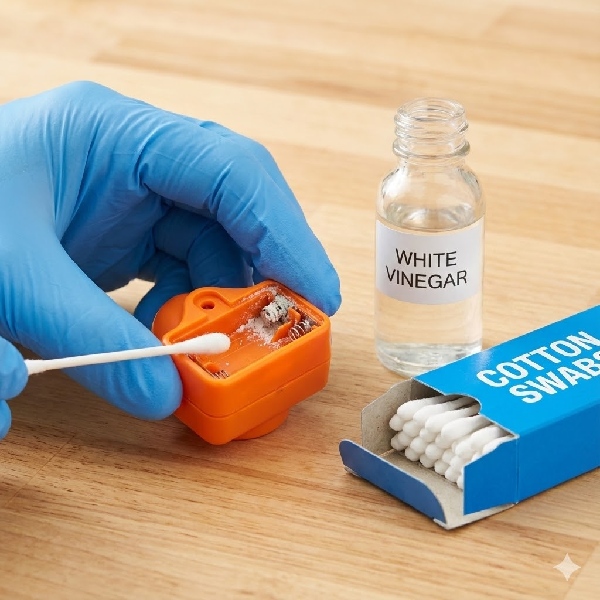
Safety First: Gear Up
The leaking fluid is caustic. It can burn your skin and irritate your eyes.
Wear Gloves: Latex or nitrile gloves are essential.
Eye Protection: Glasses or goggles are a smart move.
Ventilation: Do this in a well-ventilated room.
The Chemistry Hack: Vinegar vs. Baking Soda
To clean the corrosion, you need to neutralize the chemical.
For Alkaline Batteries (Most Common): The leakage is a base (alkaline). To neutralize a base, you need an acid. Dip a cotton swab in lemon juice or white vinegar. When you touch the white crust, it will fizz—that means it's working! Gently scrub the contacts until they are shiny metal again.
For Carbon-Zinc Batteries: The leakage is acidic. To neutralize an acid, you need a base. Mix a little baking soda and water into a paste and scrub the contacts.
Once cleaned, wipe the area with a slightly damp cloth and let it dry completely before putting in fresh batteries.
Prevention: How to Stop Leaks Before They Start
The best way to clean corrosion is to never have it in the first place. Here are three simple rules to live by:
1. The "Seasonal Swap": Do not leave batteries in seasonal devices. If you are putting away the Christmas decorations, the camping gear, or the AC remote for the winter, take the batteries out.
2. Don't Mix and Match: Never mix old batteries with new ones, or Alkaline with Carbon-Zinc. This causes uneven discharge and pushes the weaker battery to failure/leakage.
3. Store Correctly: Keep your spare batteries in a cool, dry place. Heat is the enemy of battery seals.
The Hi-Watt Difference: Engineering Against Leaks
At Hi-Watt, we understand that a battery is only as good as its casing. Whether you are a parent buying for your home or a procurement officer sourcing for a global brand, you need reliability.
We utilize advanced Anti-Leakage Technology in our manufacturing process.
Reinforced Steel Cans: Unlike cheap generic brands, our Alkaline series uses high-grade steel jackets that resist swelling.
Double-Sealing Rings: We implement specialized sealing rings at the negative terminal to withstand internal pressure variations.
0% Mercury & Cadmium: Our environmentally friendly composition isn't just good for the planet; the stable chemistry reduces the risk of volatile reactions inside the cell.
When you choose Hi-Watt, you aren't just buying energy; you are buying insurance for your electronic devices.
Final Thoughts: Don't Let a Cheap Battery Cost You an Expensive Device
It is a painful irony: saving a few cents on low-quality batteries can cost you hundreds of dollars in ruined electronics. Battery leakage is a chemical reality, but it is largely preventable. By choosing high-quality, leak-resistant batteries from a trusted manufacturer like Hi-Watt, and following proper storage habits, you can keep your gadgets running smoothly for years.
Need reliable, leak-proof power for your business or home? Explore Hi-Watt’s full range of Alkaline and Carbon-Zinc batteries today and experience the difference quality makes.





